Tofenacin
In the article below, the topic of Tofenacin will be discussed in detail and exhaustively. Tofenacin is a topic that has generated great interest in different areas, and its relevance has been increasing in recent years. Throughout this article, various aspects related to Tofenacin will be analyzed, from its origin and evolution to its implications in current society. A tour will be made of the different perspectives and approaches that have been proposed around Tofenacin, in order to offer a comprehensive and updated vision on this topic. In addition, the challenges that Tofenacin currently poses will be examined, and possible strategies and solutions will be offered to address them effectively.
 | |
| Clinical data | |
|---|---|
| Trade names | Elamol, Tofacine, Tofalin |
| Other names | tofenacin hydrochloride (USAN US) |
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| ECHA InfoCard | 100.035.746 |
| Chemical and physical data | |
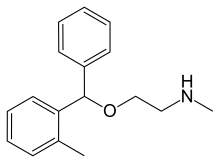
| Formula | C17H21NO |
| Molar mass | 255.361 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Tofenacin is an antidepressant drug with a tricyclic-like structure which was developed and marketed in the United Kingdom and Italy in 1971 and 1981, respectively, by Brocades-Stheeman & Pharmacia (now part of Astellas Pharma). It acts as a serotonin-norepinephrine reuptake inhibitor, and based on its close relation to orphenadrine, may also possess anticholinergic and antihistamine properties. Tofenacin is also the major active metabolite of orphenadrine and likely plays a role in its beneficial effects against depressive symptoms seen in Parkinson's disease patients.
See also
References
- ^ Index Nominum 2000: International Drug Directory. Taylor & Francis US. 2000. p. 1041. ISBN 978-3-88763-075-1. Retrieved 19 May 2012.
- ^ Buckingham J (1996). Dictionary of organic compounds: Chemical Abstracts Service registry number index. CRC Press. p. 6074. ISBN 978-0-412-54090-5. Retrieved 19 May 2012.
- ^ Pharmaceutical Manufacturing Encyclopedia (3rd ed.). Elsevier. 2013-01-15. p. 3268. ISBN 978-0-8155-1526-5. Retrieved 19 May 2012.
- ^ van Dijk J, Hartog J, Hillen FC (1 January 1978). "Non-Tricyclic Antidepressants". In Ellis GP, West GB (eds.). Progress in Medicinal Chemistry. Elsevier. p. 284. ISBN 978-0-7204-0655-9. Retrieved 19 May 2012.
- ^ Capstick N, Pudney H (1976). "A comparative trial of orphenadrine and tofenacin in the control of depression and extrapyramidal side-effects associated with fluphenazine decanoate therapy". The Journal of International Medical Research. 4 (6): 435–440. doi:10.1177/030006057600400610. PMID 800383. S2CID 40083513.
- ^ Altamura AC, Mauri MC, De Novellis F, Percudani M, Vampini V (November 1989). "Residual neuroleptic-induced parkinsonian symptoms in schizophrenia. A naturalistic study with orphenadrine". Pharmacopsychiatry. 22 (6): 246–249. doi:10.1055/s-2007-1014608. PMID 2616635. S2CID 39693625.