Aminal
In this article, we will explore Aminal from a new and detailed perspective, analyzing its various aspects and characteristics to offer a complete and enriching vision on this topic. Throughout the next few lines, we will delve into its origins, historical evolution and its relevance today, as well as examine its possible implications in different contexts. Through an exhaustive analysis, we aim to shed light on Aminal and provide new and fascinating perspectives that invite us to reflect and delve deeper into this topic.

In organic chemistry, an aminal or aminoacetal is a functional group or type of organic compound that has two amine groups attached to the same carbon atom: −C(NR2)(NR2)−. (As is customary in organic chemistry, R can represent hydrogen or an alkyl group). A common aminal is bis(dimethylamino)methane, a colorless liquid that is prepared by the reaction of dimethylamine and formaldehyde:
- 2 (CH3)2NH + CH2O → [(CH3)2N]2CH2 + H2O
Aminals are encountered in, for instance, the Fischer indole synthesis. Several examples exist in nature.
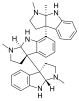
- Naturally occurring aminals
-
Hodgkinsine, an alkaloid with antiviral, antibacterial and antifungal effects
-
5,10-Methylenetetrahydrofolate, an intermediate in one-carbon metabolism
Hexahydro-1,3,5-triazine ((CH2NH)3), an intermediate in the condensation of formaldehyde and ammonia, tends to degrade to hexamethylene tetraamine.
Cyclic aminals can be obtained by the condensation of a diamine and an aldehyde. Imidazolidines are one class of these cyclic aminals.
See also
References
- ^ IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "aminals". doi:10.1351/goldbook.A00270
- ^ Gaudry, Michel; Jasor, Yves; Khac, Trung Bui (1979). "Regioselective Mannich Condensation with Dimethyl(Methylene)ammonium Trifluoroacetate: 1-(Dimethylamino)-4-methyl-3-pentanone". Org. Synth. 59: 153. doi:10.15227/orgsyn.059.0153.
- ^ Duhamel, Lucette (1982). "Aminals". In Saul Patai (ed.). Amino, Nitroso and Nitro Compounds and Their Derivatives: Vol. 2. PATAI'S Chemistry of Functional Groups. pp. 849–907. doi:10.1002/9780470771679.ch5. ISBN 9780470771679.
- ^ Hiersemann, M. "Functions bearing two nitrogens" in Comprehensive Organic Functional Group Transformations II 2005, volume 4, 411-441. Edited by Katritzky, Alan R.; Taylor, Richard J. K. doi:10.1016/B0-08-044655-8/00075-1